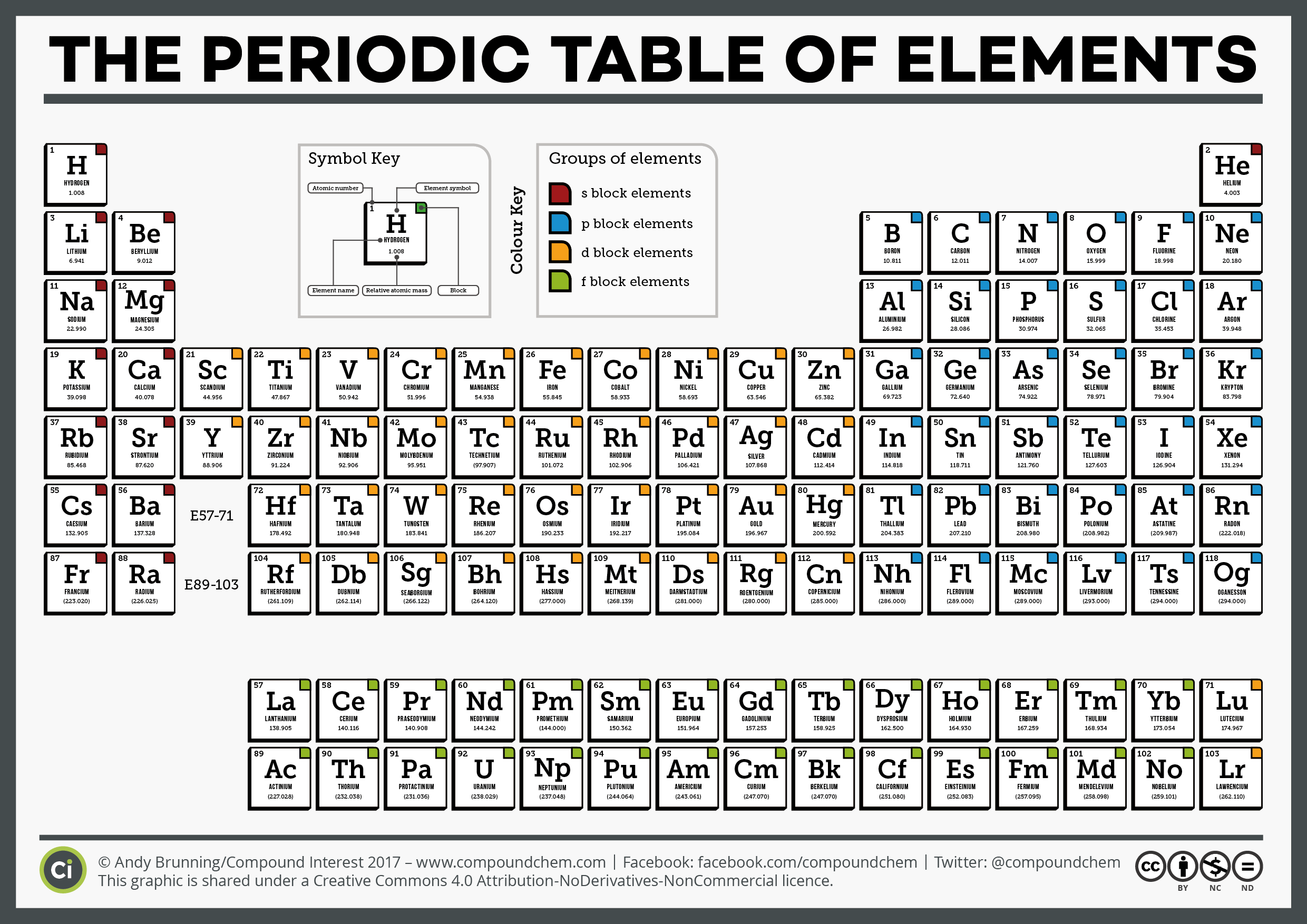
 + + - + + + + + - + - + + + + + - + + - Atoms with Few Electrons in their Outer Energy Level Notice: only 1 electron in outer level Notice: only 2 electrons in outer level ductile, good conductors, malleable, shiny, most are solid room temperature Image taken from: What metal is not a solid room temperature?.Have few electrons in their outer energy level, thus lose electrons easily Physical Properties.Found on the left of the zigzag line/staircase on the periodic table (exception Hydrogen) Chemical Properties.Sample X-ray Spectra Image taken from: ģ Classes of Elements Using this as a guide, color code your periodic table to show the three classes.He concluded that the number of protons in an atom is its atomic number.In 1914, his work led to a revision of the periodic table by rearranging the elements by their atomic numbers.“Ekasilicon” Germanium Notice how Mendeleev’s predictions (orange column) were very accurate when compared to Germanium’s actual characteristics (green column) Although Mendeleev’s Periodic Table of Elements had missing elements or “gaps,” he was able to predict the characteristics of these missing elements because of Periodic Law.Mendeleev’s Published Periodic Table of Elements Why do you think there are question marks here? Image taken from: Draft of Mendeleev's Periodic Table Image taken from:.He did this by grouping elements together according to their similarities.In the 1860’s he devised a periodic table where the elements were ordered by their atomic masses.25 were synthesized (made) by scientists Element Song. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
